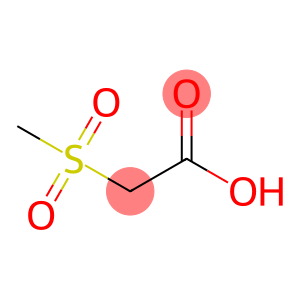
Methanesulfonylacetic Acid
Methanesulfonylacetic Acid
CAS: 2516-97-4
Molecular Formula: C3H6O4S
Methanesulfonylacetic Acid - Names and Identifiers
Methanesulfonylacetic Acid - Physico-chemical Properties
| Molecular Formula | C3H6O4S |
| Molar Mass | 138.14 |
| Density | 1.474 |
| Melting Point | 118-120°C |
| Boling Point | 423.6±28.0 °C(Predicted) |
| Water Solubility | Soluble in water. |
| BRN | 1760342 |
| pKa | pK1:2.36 (25°C) |
| Storage Condition | Sealed in dry,Room Temperature |
| MDL | MFCD00051843 |
Methanesulfonylacetic Acid - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R36 - Irritating to the eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
Methanesulfonylacetic Acid - Reference Information
| Use | methanesulfonylacetic acid is a raw material for the synthesis of pharmaceuticals and agrochemicals, which is used as a synthetic herbicide, the use of intermediates for antibiotics and bleach activators is particularly important. Methanesulfonyl Group is a group often introduced in the synthesis of pharmaceutical and chemical intermediates, and has a variety of biological activities. |
| preparation | 4-methylthioethane is used as raw material, which is firstly chloromethylated to produce 4-methylthiochloroethane, magnesium is then added to form a Grignard reagent, hydrolyzed with carbon dioxide to give 4-methylthioacetic acid, and finally oxidized to 4-methanesulfonylacetic acid. In the synthetic route, the Grignard reagent reaction requires high requirements, and must be anhydrous and oxygen-free, and the experimental operation requirements are also high, thus increasing the difficulty of industrialization. In this paper, 1-chloro-2-(methylthio) ethane is used as raw material, and 4-methylthioacetic acid is obtained by hydrolysis after cyano nucleophilic substitution, and finally oxidized to methylsulfonyl acetic acid. Its synthetic route is shown in Figure 1. In the synthetic route, sodium hydride has serious toxic environmental pollution, more nucleophilic substitution by-products, and higher reaction operation requirements, which is not conducive to industrial production and environmental friendliness. |
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: METHANESULFONYLACETIC ACID Visit Supplier Webpage Request for quotationCAS: 2516-97-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Methanesulfonylacetic acid Visit Supplier Webpage Request for quotationCAS: 2516-97-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: Methanesulfonylacetic Acid Request for quotation
CAS: 2516-97-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2516-97-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: METHANESULFONYLACETIC ACID Request for quotation
CAS: 2516-97-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2516-97-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Methylsulfonylacetic Acid Visit Supplier Webpage Request for quotationCAS: 2516-97-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: METHANESULFONYLACETIC ACID Visit Supplier Webpage Request for quotationCAS: 2516-97-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Methanesulfonylacetic acid Visit Supplier Webpage Request for quotationCAS: 2516-97-4
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: Methanesulfonylacetic Acid Request for quotation
CAS: 2516-97-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 2516-97-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: METHANESULFONYLACETIC ACID Request for quotation
CAS: 2516-97-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2516-97-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Methylsulfonylacetic Acid Visit Supplier Webpage Request for quotationCAS: 2516-97-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




